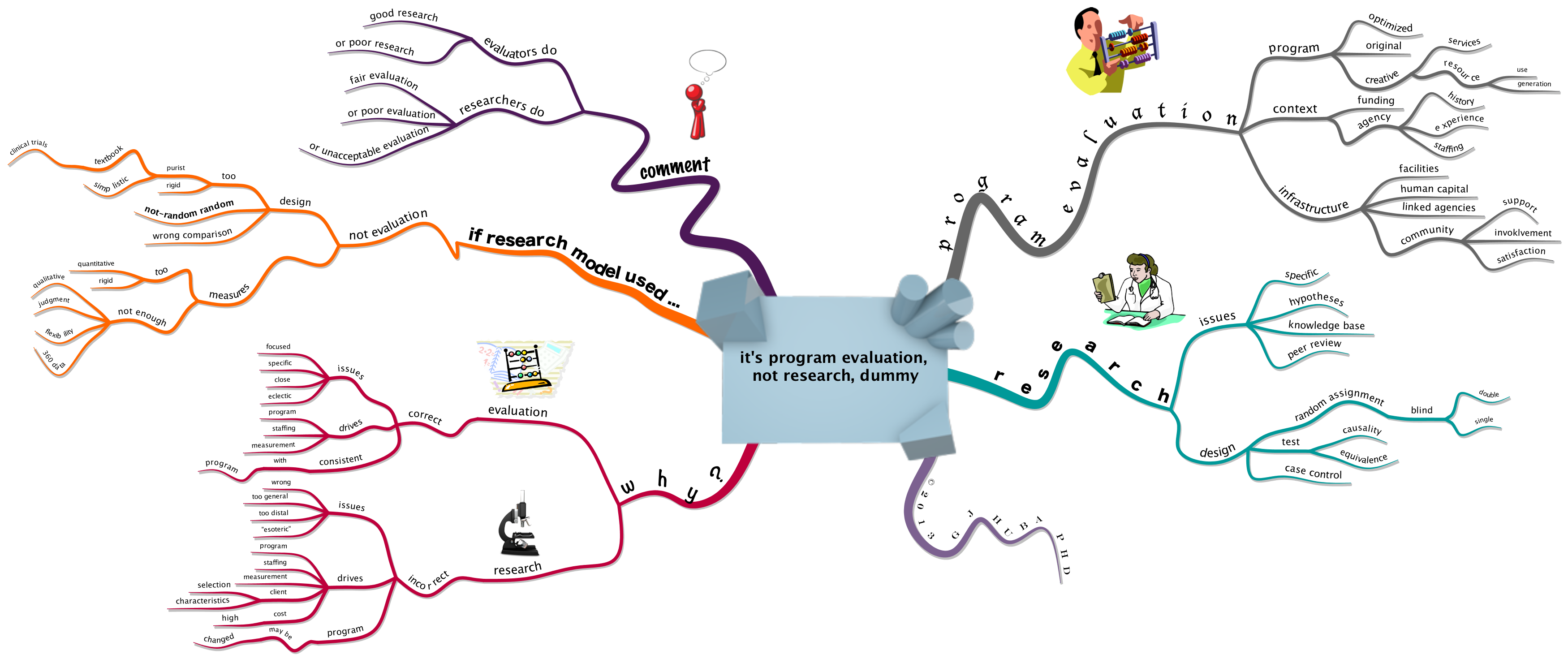
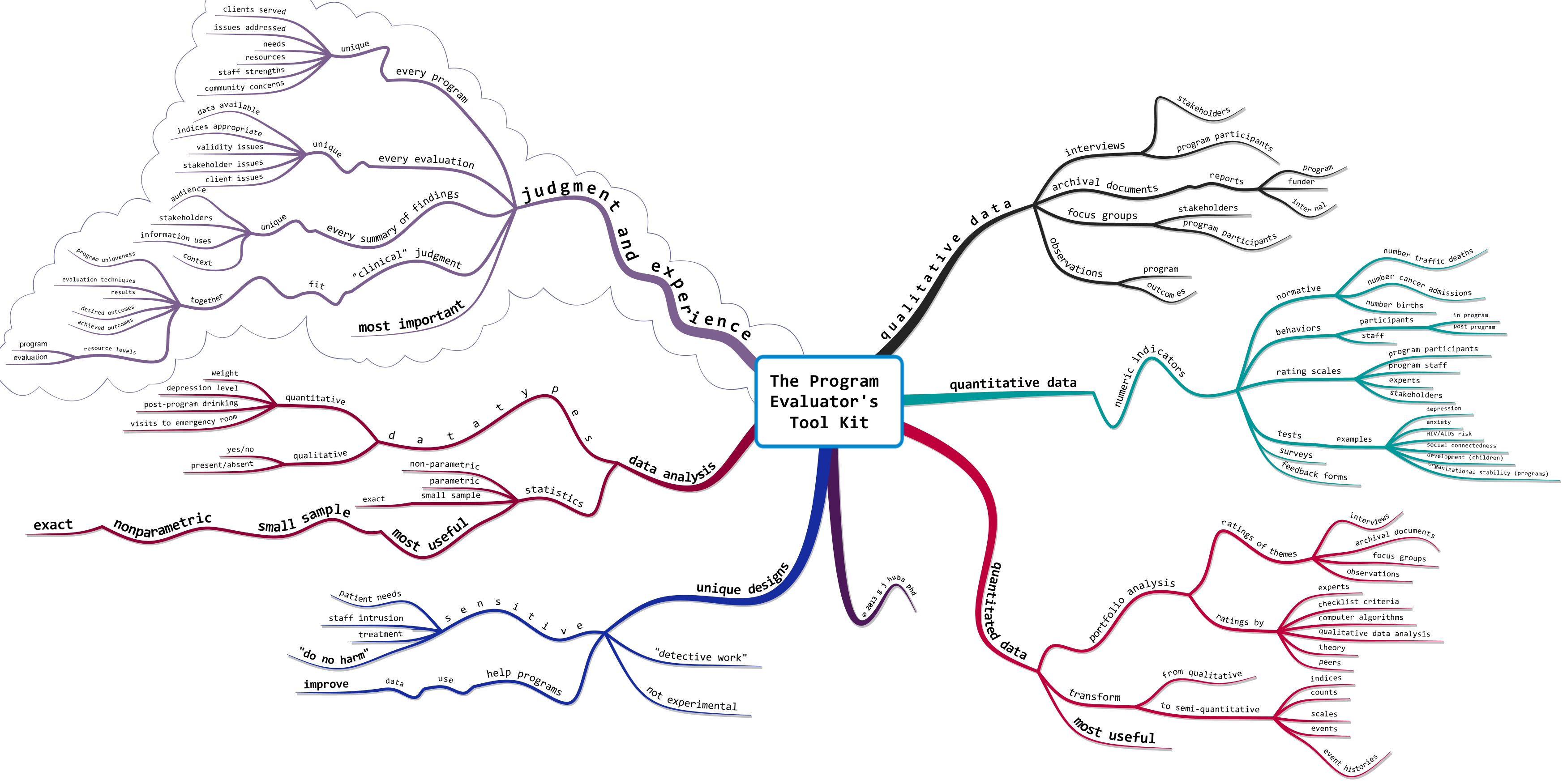
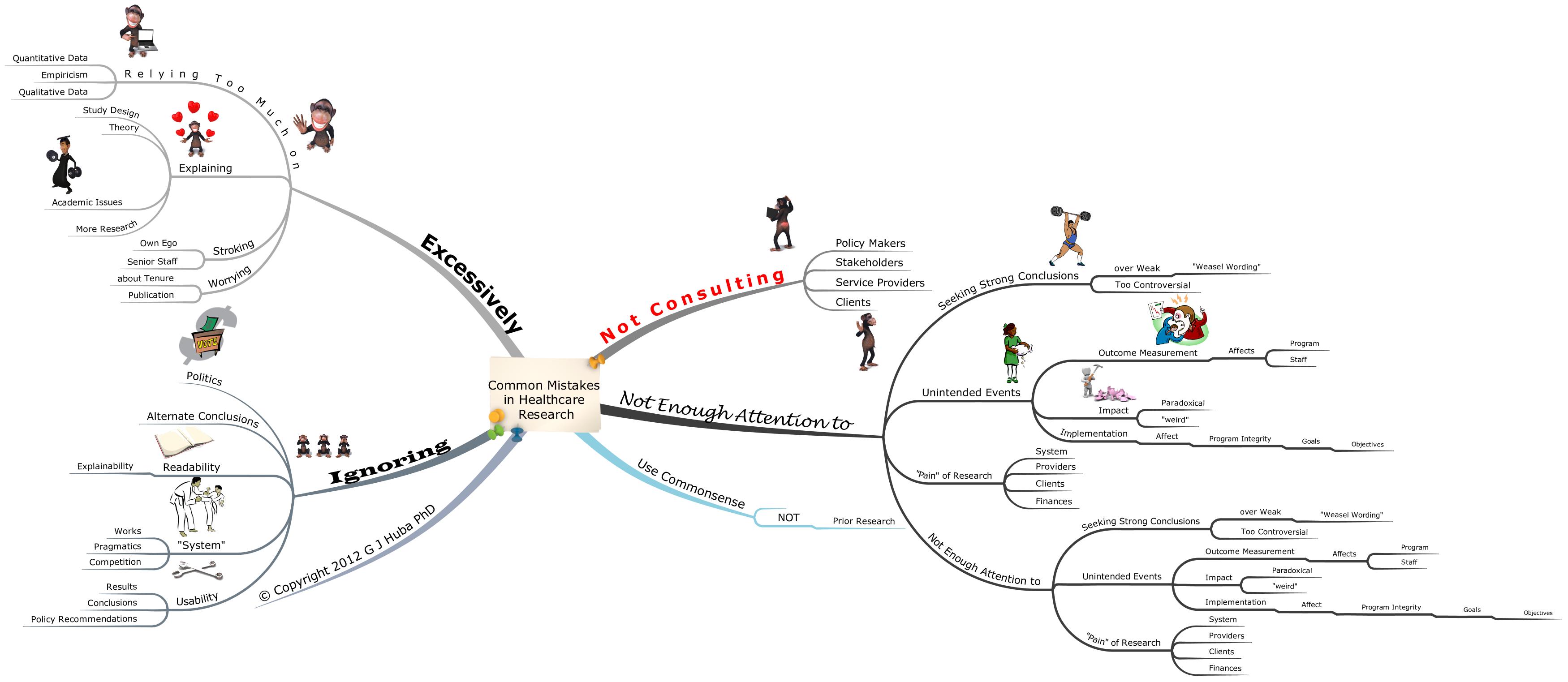
Remember the “gold standard” research paradigm for determining if a medical treatment works: the DOUBLE BLIND, RANDOM ASSIGNMENT EXPERIMENT?
The design has historically been considered the best way to “prove” that new medical interventions work, especially if the experiment is replicated a number of times by different research teams. By the double blind (neither the treating medical team nor the patient know whether the patient is taking a placebo or active medication) design, investigators expect to negate the placebo effects caused by patient or medical staff beliefs that the “blue pill” is working.
A key part of virtually all double-blind research designs is the assumption that all patient expectations and reports are independent. This assumption is made because of the statistical requirements necessary to determine whether a drug has had a “significantly larger effect” as compared to a placebo. Making this assumption has been a “standard research design” feature since long before I was born more than 60 years ago.
2013
Google the name of a new drug in clinical trials. You will find many (hundreds, thousands) of posts on blogs, bulletin boards for people with the conditions being treated with the experimental drug, and social media, especially Twitter and Facebook. Early in most clinical trials participants start to post and question one another about their presumed active treatment or placebo status and whether those who guess they are in the experimental condition think the drug is working or not. Since the treatments are of interest to many people world-wide who are not being treated with effective pharmaceuticals, the interest is much greater than just among those in the study.
Google the name of a new drug being suggested for the treatment of a rare or orphan disease that has had no effective treatments to date and you will find this phenomenon particularly prevalent for both patients and caregivers. Hope springs eternal (which it SHOULD) but it also can effect the research design. Obviously data that are “self reported” from patient or caregiver questionnaires can be affected by Internet “the guy in Wyoming says” or the caregiver of “the woman in Florida.”
OK you say, but medical laboratory tests and clinical observations will not be affected because these indices cannot be changed by patient belief they are in the experimental or placebo conditions. Hhmmm, Sam in Seattle just posted that he thinks that he in the experimental condition and that his “saved my life” treatment works especially well if you walk 90 minutes a day or take a specific diet supplement or have a berry-and-cream diet. Mary in Maine blogs the observation that her treatment is not working so she must be in the placebo condition and becomes very depressed and subsequently makes a lot of changes in her lifestyle, often forgetting to take the other medications she reported using daily before the placebo or experimental assignment was made.
Do we have research designs for the amount of research participant visible (blogs, tweets, bulletin boards) and invisible (email, phone) communication going on during a clinical trial? No. Does this communication make a difference in what the statistical tests of efficacy will report? Probably. And can we ever track the invisible communications going on by email? Note that patients who do not wish to disclose their medical status will be more likely to use “private” email than the public blog and bulletin board methods.
Want an example. Google davunetide. This was supposed to be a miracle drug for the very rare neurodegenerative condition PSP. The company (Allon) that developed the drug received huge tax incentives in the USA to potentially market an effective drug for a neglected condition. The company, of course, was well aware that after getting huge tax incentives to develop the pharmaceutical, if the drug were to prove effective in reducing cognitive problems (as was thought), it would then be used with the much more common (and lucrative from the standpoint of Big Pharma) neurodegenerative disorders (Alzheimer’s, Parkinson’s) and schizophrenia.
Patients scrambled to get into the trial because an experimental medication was better than no medication (as was assumed, although not necessarily true) and the odds were 50/50 of getting the active pills.
Patients and caregivers communicated for more than a year, with the conversations involving patients from around the world. In my opinion, the communications probably increased the placebo effect, although I have no data nor statistical tests of “prove” this and it is pure conjecture on my part.
The trial failed miserably. Interestingly, within a few weeks after announcing the results, the senior investigators who developed and tested the treatment had left the employ of Allon. Immediately after the release of the results, clinical trial participants (the caregivers more than the patients) started trading stories on the Internet.
Time for getting our thinking hats on. I worked on methodological problems like this for 30+ years, and I have no solution, nor do I think this problem is going to be solved by any individual. Teams of #medical, #behavioral, #communication, and #statistical professionals need to be formed if we want to be able to accurately assess the effects of a new medication.
Click on the image to expand.

Like this:
Like Loading...



 I consider John W. Tukey to be the King of Little Data. Give him a couple of colored pencils, the back of a used envelope, and some data and he could bring insight to what you were looking at by using graphic displays, eliminating “bad” data, weighting the findings, and providing charts that would allow you to explain what you were seeing to those who had never been trained in technical fields.
I consider John W. Tukey to be the King of Little Data. Give him a couple of colored pencils, the back of a used envelope, and some data and he could bring insight to what you were looking at by using graphic displays, eliminating “bad” data, weighting the findings, and providing charts that would allow you to explain what you were seeing to those who had never been trained in technical fields.